
Examples of Neutral Atom- 118 Elements of the Periodic Table. There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. In that spirit, the present contribution is an attempt to look back and review our understanding of the development of the periodic system of the chemical elements, with a particular emphasis on the question as to why the chemical elements exist either as metals or non-metals under ambient conditions on Earth. Please note that the elements do not show their natural relation towards each other as in the Periodic system. Therefore, all the elements in the periodic table are neutral atoms. The first chemical element is Actinium and the last is Zirconium. You can also install the most recent version from the repository: pip install git+įull documentation is hosted on Read the Docs. As a result, the atom’s total electric charge is zero, and it is said to be neutral. 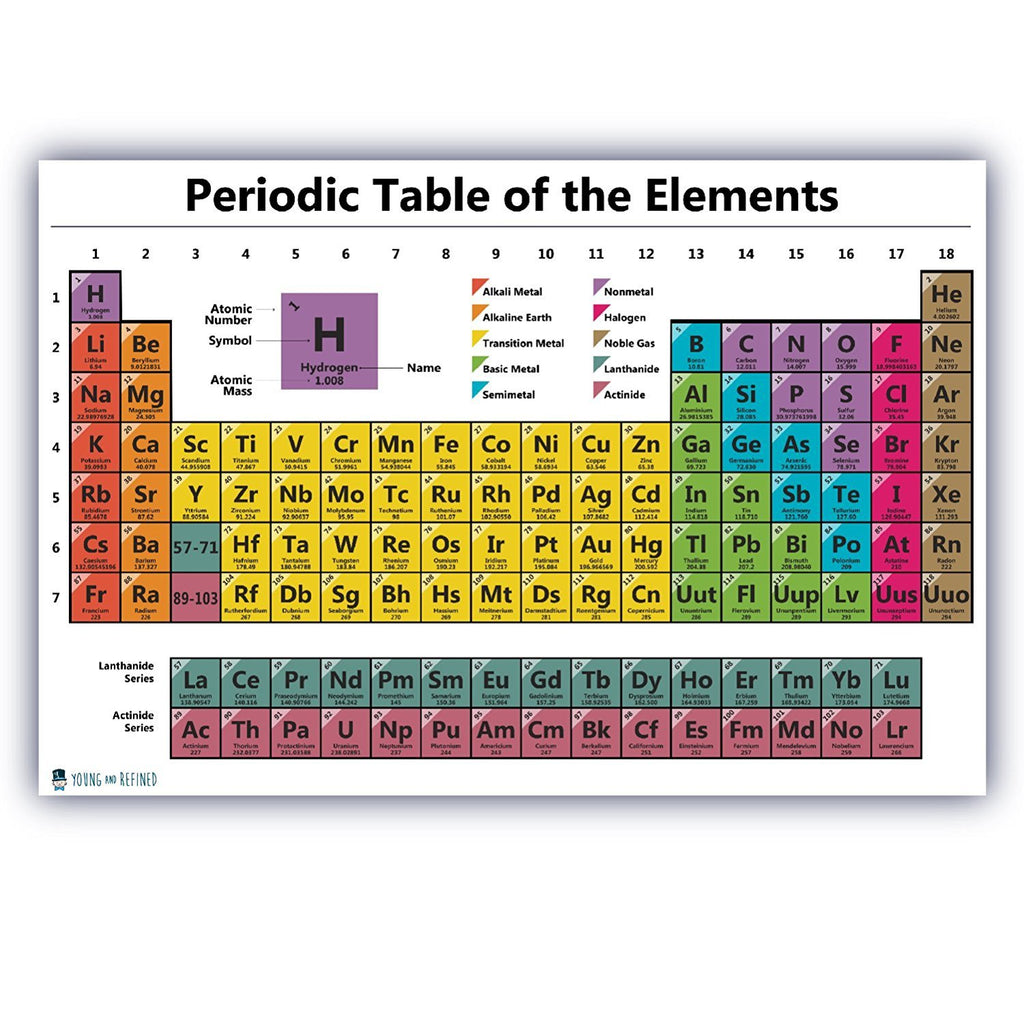
The preferred installation method is withĬhannel by conda install -c lmmentel mendeleev=0.14.0 Relations between various properties of elements. Where you can create your own periodic tables and visualize the Nonmetallic chemical element with symbol C from Latin carbo for coal, with atomic number 6. If you would like to explore the data available in If you want toĪvailable as jupyter notebooks. You could help by writing documentation, pull-requests, report issues and/or translations. It is development and maintained by developers in an Open Source manner. Moreover it provides an easy to use interface toĮnables you to create customized periodic tables displaying variousĭjango Extensions is free and always will be. Properties of elements, ions and isotopes in the periodic table of This package provides a convenient python API for accessing various Of elements, ions and isotopes in the periodic table of elements. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.A package with a convenient python API for accessing various properties Here are facts about this important and interesting element. This nonmetallic element is the key to the chemistry of living organisms, primarily due to its tetravalent state, which allows it to form four covalent chemical bonds with other atoms. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Carbon is the element with atomic number 6 on the periodic table with symbol C. It has low density, is non-toxic, has a high thermal conductivity, has excellent corrosion resistance and can be easily cast, machined and formed. This is because of its particular properties. Free Gift for you: Interactive Periodic Table Aluminium is used in a huge variety of products including cans, foils, kitchen utensils, window frames, beer kegs and aeroplane parts. It is a group IVA element, residing between boron and nitrogen on the periodic table, and it has 6 protons, 6 neutrons, and 6 electrons. Metals reside on the left side of the table, while non-metals. In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. The periodic table (also known as the periodic table of elements) is organized so scientists can quickly discern the properties of individual elements such as their mass, electron number, electron configuration and their unique chemical properties. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. The Chemistry Divisions Periodic Table describes the history, properties, resources, uses, isotopes, forms, costs, and other information for each element. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
